Guest Author: Rauful Alam
Writer and Researcher
Email: [email protected]
In this edition, we will primarily discuss electrophiles and nucleophiles. Incidentally, some other related topics will be included as well. What is an electrophile? The name comes from an affinity for electrons, or electrophilicity. That is, any molecule or part of a molecule (such as a functional group) that is eager to accept electrons is generally called an electrophile. In other words, an electrophile is an electron acceptor. The electron density in an electrophile is low. That’s why it has an affinity for electrons. In organic compounds, the carbon atom of a compound or group is usually the electrophile. That is, if a certain carbon atom has low electron density, it will want to accept electrons. By accepting electrons, new bonds are formed.
The opposite of an electrophile is a nucleophile. If an electrophile is an electron acceptor, then a nucleophile is an electron donor. Who can donate electrons? — Certainly one that has excess electrons or has a low ability to retain electrons.
When we express this generally, it’s shown as follows. An arrow is used to indicate the nucleophile attacking the electrophile, or donating a pair of electrons.
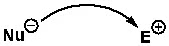
To understand a reaction mechanism, it is very important to identify the electrophile and nucleophile. There are several easy rules for this identification, which will be discussed sequentially.
Identifying nucleophiles is comparatively easy. Alcohols, amines, thiols—these are nucleophiles. This is because oxygen, nitrogen, and sulfur atoms contain lone pairs or free pairs of electrons. Below are some examples of nucleophiles. The oxygen, nitrogen, and sulfur atoms in these molecules have electron pairs or lone pairs. You should be able to figure out how many lone pairs each has with a bit of thought. This has been discussed in earlier parts.

Besides these, there are other nucleophiles as well, especially carbon nucleophiles. These will appear and be revisited in future parts.
Previously we learned about aldehyde and ketone groups. Aldehydes and ketones have a carbon-oxygen double bond, which we call a carbonyl. In the carbonyl group, oxygen is more electronegative than carbon, and there is a carbon-oxygen double bond. Thus, the carbonyl is an electrophile—specifically, the carbon atom in the carbonyl group has lower electron density, making it an electrophile.
This raises the question: alcohols also have a carbon-oxygen bond. Are alcohols electrophiles? No, alcohols are not electrophiles. Or in other words, alcohols are not considered electrophiles. Rather, alcohols are considered nucleophiles, since they have a single carbon-oxygen bond.
Therefore, electrophilicity doesn’t depend on electronegativity alone. The molecular geometry, the presence of double or triple bonds, bond length, and even the stability of the part of the molecule acting as the electrophile after disconnection—all these factors are involved. These aspects will be discussed with various reaction examples.
Electrophiles can also be categorized as strong or weak electrophiles. We will understand these concepts more easily through examples. For instance, alkyl halides are electrophiles. Due to the carbon-halogen bond, the electron density at the carbon atom is comparatively lower.
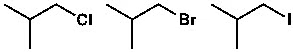
As shown above, three isobutyl molecules are given as examples. Among these, isobutyl iodide is a strong or better electrophile. That is, it will readily react with a nucleophile. The nucleophile can easily replace iodine, breaking the carbon-iodine bond. Chlorine, bromine, and iodine—all three have greater electronegativity than carbon. However, the carbon-iodine bond length is longer—or greater—than that of carbon-chlorine or carbon-bromine. If a single bond is longer, it is weaker. And weaker bonds break more easily. As a result, the carbon-iodine bond in isobutyl iodide will break more easily. That’s why it’s called a strong electrophile.
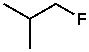
On the other hand, fluorine has the highest electronegativity. By that logic, the carbon-fluorine bond should be the strongest electrophile. But it’s not. As mentioned earlier, electronegativity alone does not determine electrophilicity. Because fluorine is highly electronegative, the C—F bond is very strong. Breaking this bond requires a lot of energy, known as bond dissociation energy (BDE); for example, the BDEs of C—F, C—Cl, C—Br, and C—I are 110, 84, 72, and 57 kilocalories/mole respectively. You don’t have to memorize these numbers—just remember which bonds are strong and which are comparatively weaker.
Another important concept here is the leaving group ability. The group that gets detached in a reaction is called the leaving group. For example, in the previous example, Cl, Br, I—these are leaving groups. When the nucleophile attacks—or donates a pair of electrons—the halogen atom leaves as an anion.

The example above will clarify the concept further (the full reaction is not shown). Thiol is the nucleophile here, and isobutyl iodide is the electrophile. When illustrating, the first arrow (blue) is drawn from the nucleophile to the electrophile, as the thiol donates a pair of electrons; iodine thus departs with a pair of electrons. The carbon-iodine bond breaks—this is a substitution reaction. The second arrow (red) goes from the carbon-iodine bond to the iodine. These arrows essentially represent electron flow. Illustrating the mechanism this way is called the “push-pull mechanism.” And this method for demonstrating or explaining is universal. It’s important to understand and know the conventions of science’s universal language and methods.
In this example, iodine is the leaving group, since it departs. Iodine leaves as iodide (I-). If a leaving group is stable after leaving, it makes the electrophile stronger. To put it simply: if something is happier or more comfortable after leaving, it will depart more readily. The nucleophile won’t have to work too hard.
So when determining whether an electrophile is strong, the leaving group ability must also be considered. Among F, Cl, Br, I, iodine easily leaves from the carbon-iodine bond, because iodide is quite stable after dissociation. This can be understood using the logic of conjugate acids and bases.
Iodide is the conjugate base of hydroiodic acid. Since hydroiodic acid is very strong, its conjugate base will be very stable. The definition of a strong acid is how easily and quickly it can donate a proton or hydrogen ion.
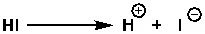
In the same way, fluoride (F-) is less stable than iodide. So as a leaving group, iodine is excellent—a good leaving group. Fluorine is a poor leaving group.
Now let’s discuss aromatic compounds as electrophiles. Compared to aliphatic compounds, various additional considerations are necessary to explain their properties.
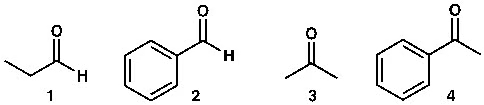
The first molecule is an aliphatic aldehyde; the second is an aromatic aldehyde (benzaldehyde). Which of these two is the stronger electrophile? In other words, which will react more quickly with a nucleophile?
The answer is the first molecule. Why?
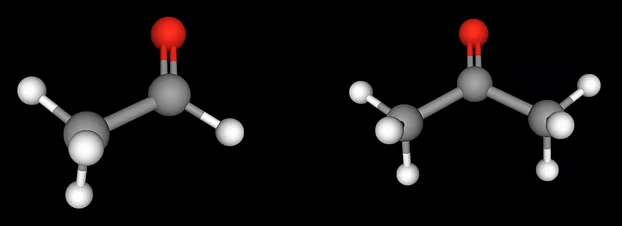
Because the aromatic ring has pi electrons—six conjugated pi electrons. The pi bond in the carbonyl group also involves two pi electrons. The pi electrons of the benzene ring and the pi electrons of the carbonyl group are conjugated (see the image below), creating stable conjugation. As a result, the electron density on the carbonyl carbon in aromatic aldehydes is higher than that of aliphatic compounds. To put it simply, that’s how it works—but in reality, this can also be explained differently using molecular orbital theory, which is for advanced levels.
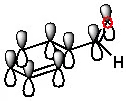
The third molecule, acetone, is a weaker electrophile than the first. This is because, in aldehydes, the carbonyl group is attached to a hydrogen atom. In ketones, that hydrogen is replaced by a carbon, so the carbonyl carbon is more “shielded.” That is, there is greater steric hindrance, making it harder for the nucleophile to attack. The image below gives an idea of the steric difference between aldehydes and ketones.
The fourth molecule, acetophenone, is again a weaker electrophile than acetone. Here too, the benzene ring’s pi electrons are conjugated with the carbonyl’s pi electrons.
Since carbonyl compounds are very important electrophiles, it’s good to remember that, in general, aliphatic aldehydes are stronger electrophiles than aromatic aldehydes. Likewise, aldehydes are stronger electrophiles than ketones.
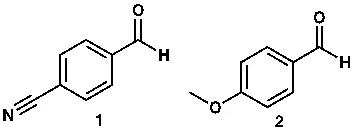
The electrophilicity of aromatic aldehydes or ketones is also influenced by the functional groups attached to the aromatic ring. For example, among the two molecules below, the first is a stronger or better electrophile because the benzene ring has a nitrile (R–CN) group attached. The nitrile group is an electron-withdrawing group (EWG)—it pulls electrons away. This decreases the electron density at the carbonyl carbon. The opposite happens in the second molecule: since methoxy is an electron-donating group (EDG), the electron density on the carbonyl carbon is comparatively higher.
The next installment will discuss more varied classes of electrophiles and nucleophiles.
References:
This article was adapted from a feature published in Bigganchinta Magazine.
Author: Dr. Rauful Alam, Researcher, University of Pennsylvania, USA.











Leave a comment