Rauful Alam
Researcher, University of Pennsylvania, USA
You must have heard of organic compounds. Chemical molecules or compounds made up of carbon and hydrogen atoms are generally called organic compounds or organic molecules. Sometimes, they’re also called hydrocarbons, since they contain hydrogen and carbon atoms.
It’s not always just carbon and hydrogen; these two atoms are present in higher amounts or in higher ratios. However, other atoms such as oxygen, nitrogen, sulfur, etc., can also be present.
The reason they’re called organic compounds is because they are the compounds of living beings, of life itself. In other words, wherever there is life, there are organic compounds. Plants, animals, bacteria, and viruses all mainly consist of organic compounds. In the human body, countless organic compounds are broken down and formed every day. This process of breaking down and building up is called a chemical reaction. To write and represent these chemical reactions, we use chemical structures and various symbols. So, what’s the main difference between a chemical formula and a chemical structure?
The key difference is that a formula only tells you what atoms are present and in what quantities. But a chemical structure shows how these atoms are arranged in the molecule, how one atom is bonded to another. Chemical structures give us a more detailed understanding of the molecule. Let me give you an example.
Almost all of you use hand sanitizer. Many hand sanitizers contain an organic compound called isopropyl alcohol, also known as 2-propanol. Its chemical formula is C3H8O. This means the molecule contains three carbon, eight hydrogen, and one oxygen atom. The structure of the compound is shown below:
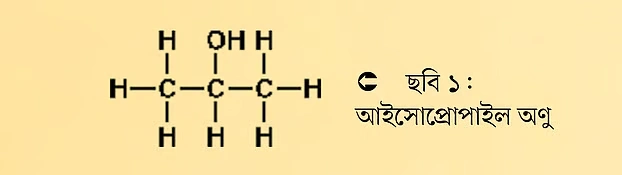
From the chemical structure, it’s immediately clear which atoms the oxygen atom is bonded to. It is connected to a carbon and a hydrogen atom. We also see that the oxygen is bonded to the central carbon atom. If it were bonded to a different carbon instead of the central one, it wouldn’t be isopropanol anymore—it would become a different molecule. So, from the chemical structure, we can gather various information. Therefore, there are specific rules for drawing chemical structures.
The practice of representing organic compounds with structures began about 200 years ago. Over time, we have developed modern ways to draw and represent organic compounds. Even though we can’t see molecules with the naked eye, we have been able to determine the structures of countless molecules using advanced instruments and research.
Those who work or conduct research with organic compounds are called organic chemists. They are very skilled in drawing organic compounds. This skill comes with learning and practice. If structures are drawn incorrectly, it becomes difficult for others to understand. That’s why organic chemists around the world follow certain rules for drawing organic compounds. This is a language—if you can illustrate a reaction properly, it conveys many things at once without detailed explanation. Besides drawing by hand, nowadays, many software tools are used, such as ChemDraw. This software is very popular among organic chemists worldwide. For the illustrations in this article, I have used it as well. Still, organic chemists also learn and practice drawing by hand.
Today, I will discuss some rules for drawing organic compounds. Even at higher levels in Bangladesh, many do not follow the correct conventions. I felt it necessary to present how organic compounds are drawn internationally. If college and university students practice this, they will benefit. If teachers adopt and encourage this practice, students will be able to learn the proper and globally accepted conventions in time.
This article will be written sequentially in several parts. First about drawing, then later about the chemical reactions of organic compounds in a simple manner.
When we write the formula of methane (also called methene), we just write CH4. It means one carbon atom is bonded to four hydrogen atoms. But if we want to represent it more deeply, we can draw it as in the picture below (Figure 2). This way, we do not only show the formula, but also give an idea about its geometric structure. It shows that the methane molecule is tetrahedral. The specific structure of molecules determines many aspects of their chemical reactivity. If we write the formula for ethane, it would be C2H6 or CH3-CH3. However, organic chemists often represent it in different ways for convenience and to save time. Time is of great importance to organic chemists.
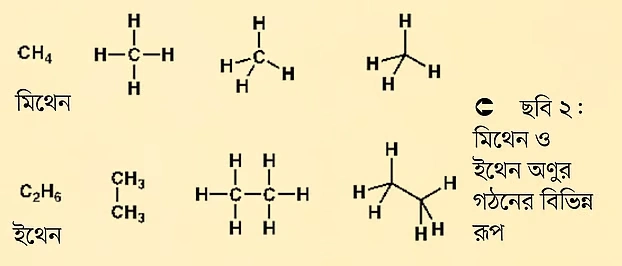
As the number of carbon atoms increases, drawing all the carbon-hydrogen bonds and structures takes a lot of time. Also, molecules are often represented linearly by convention, but in reality, molecules are not so straight or linear.
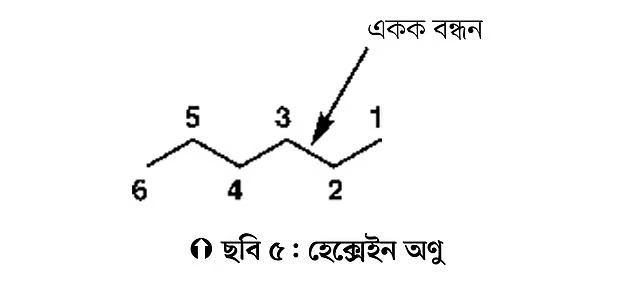
For example, propane contains three carbon atoms (see Figure 3). There are several ways to represent its chemical structure. In the modern method, propane is shown simply by drawing two straight lines joined together. The lines form an angle of about 120 degrees. The corner carbon atom in this angle is called the methylene group (CH2), and the two ends are methyl groups (CH3). Drawing this way is easy, quick, and represents the three-dimensional structure fairly well. Here, examples of butane, pentane, and hexane molecules are also given (see Figure 4). Students can practice drawing other molecules in the same way.
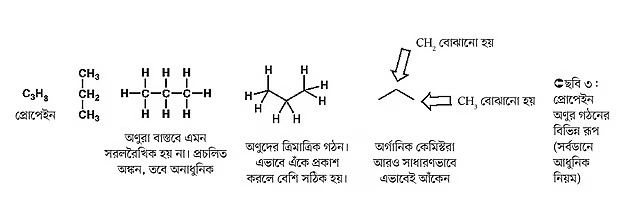
The above molecules are called saturated hydrocarbons. What does “saturated” mean? When all the carbon atoms in an organic compound are joined by single bonds, those compounds are called saturated hydrocarbons.
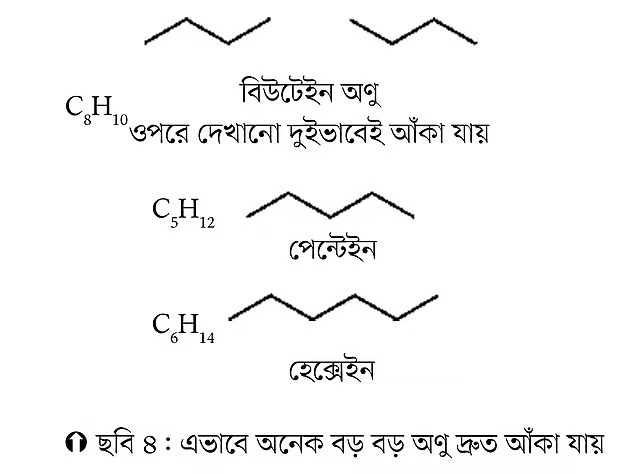
Now, let’s practice counting carbon atoms using hexane as an example. If you count the carbon atoms after drawing the hexane structure above, you get six carbon atoms. Four carbon atoms are internal (internal; 2-5) and two are terminal (terminal; 1 and 6). The terminal carbons are methyl groups (CH3), and the internal carbons are methylene groups (CH2).
The molecule examples above are all straight-chain or linear molecules. These are also called acyclic molecules. That means there are also cyclic molecules. Cyclohexane, for example, can exist as both acyclic and cyclic forms, but they are not the same molecule—they have different structures.
The molecule below is cyclohexane. The “cyclo” in the name indicates its cyclic structure.
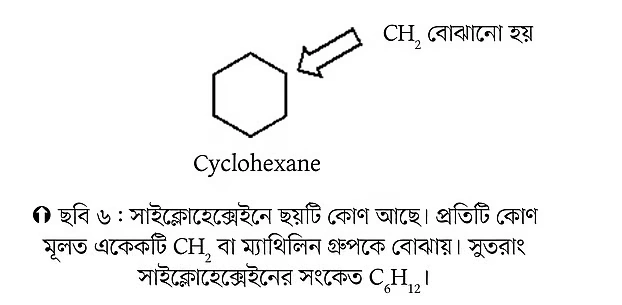
Does cyclohexane have any methyl groups? Think about it for a moment. We also know about the three-dimensional structure of cyclohexane. Cyclohexane mainly exists in a chair-like conformation, which is its most stable form. That’s why it’s called chair conformation. I won’t go into detail about chair conformation now, since we’ll discuss it later again.
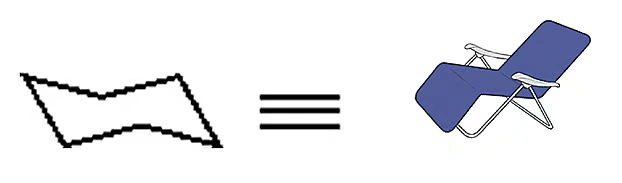
If you practice drawing a few molecules like this, the whole concept will become easier; it’ll be easier to remember. Once we learn modern chemical structure drawing, it will be much easier to understand and explain many things, including chemical reactions. Many students in Bangladesh find chemical reactions hard to understand or confusing, and one reason is a lack of a solid understanding of chemical structure. In the next part, I’ll discuss some more fascinating topics.
References:
This article is adapted from a feature published in the BigganChinta Magazine.
Author: Dr. Rauful Alam, Researcher, University of Pennsylvania, USA.









Leave a comment