Rouful Alam
Researcher, University of Pennsylvania, USA
In the previous installment, we discussed compounds with only single bonds. In this episode, we will talk about drawing molecules that contain multiple bonds and functional groups.
Compounds with Multiple Bonds
Organic compounds that contain only carbon-carbon single bonds (C–C bond) are called saturated compounds. If, besides single bonds, the compound also contains carbon-carbon double (C=C) or triple bonds, it is called an unsaturated compound. One thing to keep in mind: if a compound contains a carbon-oxygen double bond, or a carbon-nitrogen double or triple bond, that compound is not considered unsaturated. The saturated/unsaturated classification depends only on carbon-carbon bonds.
The names of compounds with a carbon–carbon double bond end in “ene”, and compounds with a triple bond end in “yne”. This makes it easier to identify the type of compound. Generally, compounds with double bonds are referred to as alkenes.
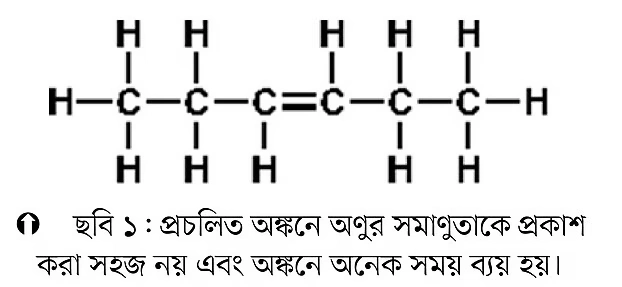
Let’s take the example of hexene. Hexene has six carbon atoms, hence “hex”, and since it has a double bond, “ene” is added at the end to make “hexene”. The most correct name for the compound, however, would be 3-hexene. This is discussed below.
If we draw hexene as in Figure 1, it takes a lot of time. Also, if the compound contains a double bond, there are two isomers. One is the trans-isomer, and the other is the cis-isomer. Trans and cis isomers are called geometric isomerism. With the old drawing method, it’s difficult to clearly represent this kind of isomerism. If we draw using the modern method as in Figure 2 below, it becomes much more understandable. Nowadays, organic chemists all over the world use this modern method for drawing. All prestigious journals and famous books follow this approach.

The trans-isomer is when the two carbon atoms closest to the double bond (marked with pink balls) are positioned far from each other. The cis-isomer is when those two carbon atoms are close to each other. With a little practice, you’ll be able to draw these isomers quickly.
Remember that geometric isomerism and chirality (optical isomerism) are not the same. Many people confuse them. Geometric isomerism has nothing to do with symmetry. For optical isomerism, the compound must be asymmetric. These topics will be discussed in detail later.
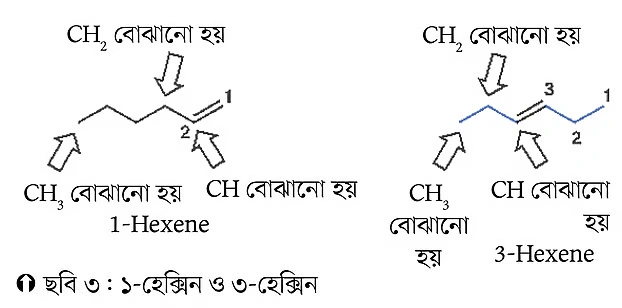
If a compound contains a double bond, its position needs to be determined. To do this, count the carbon atoms and indicate the position of the double bond with a number. The numbering starts from the end that gives the double bond the lowest number. For example, Figure 3 below demonstrates this with two examples.
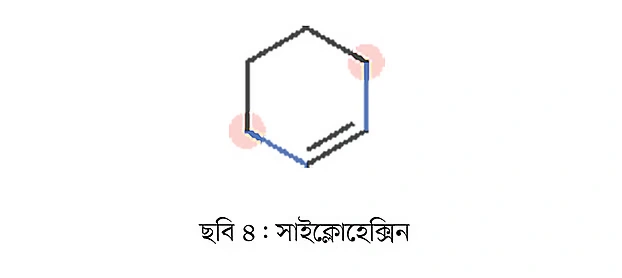
The above examples are all acyclic or linear compounds. Cyclic compounds can also contain double bonds, such as cyclohexene (Figure 4).
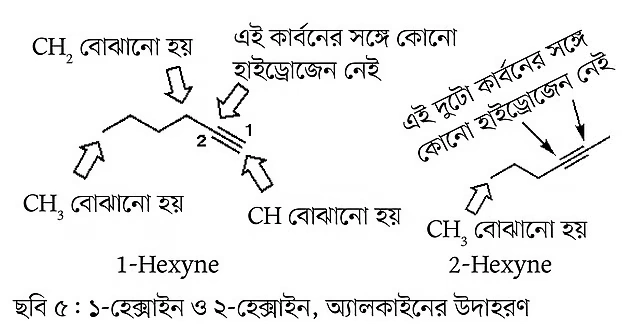
Compounds with carbon-carbon triple bonds are also found in nature. These compounds are generally called alkynes (see Figure 5). These types of compounds do not have cis and trans isomers. In alkyne compounds, there is at least one carbon atom not attached to any hydrogen atom.
Alkynes can also be cyclic. For example, cyclooctyne is a well-known example (Figure 6). This compound has eight carbon atoms.
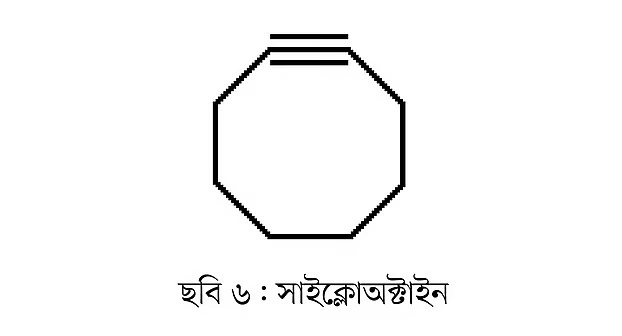
Compounds like these are very chemically reactive, meaning they react easily. As a result, their stability is low. Separating or purifying very reactive compounds is quite challenging and requires extra precautions.
All the double and triple bonded compounds presented so far are quite volatile, meaning they have low boiling points. The reason is that compounds made only of carbon and hydrogen are usually volatile unless they are very large in size or have high molecular mass. This is because the intermolecular forces in these molecules are weak. Hydrogen bonding increases intermolecular forces, but for hydrogen bonding, atoms other than carbon and hydrogen need to be present.
Although the main atoms in organic molecules are carbon and hydrogen, oxygen, nitrogen, and sulfur atoms are also found. Organic chemists call these “heteroatoms”. However, the term “heteroatom” usually refers mainly to nitrogen.
Oxygen can appear in organic compounds as different functional groups, such as alcohols, ethers, aldehydes, ketones, etc. Aldehydes and ketones are collectively called carbonyl groups. A carbonyl group is a carbon-oxygen double bond.
If an organic compound contains a hydroxy group (OH), it is generally called an alcohol. The names of such compounds end with “ol”, for example: methanol, ethanol, isopropanol, and tertiary-butanol or t-butyl alcohol. These alcohols have low molecular mass and are widely used. For instance, these compounds are used as solvents all over the world. Ethanol is used as a fuel for vehicles. As a beverage, ethanol is used. Isopropanol or IPA is very popular as a solvent. Currently, about 2-2.5 million metric tons of IPA are produced annually worldwide. IPA is used in hand sanitizers.

Alcohols with more carbon atoms are also widely used. These compounds are utilized in various chemical reactions. In many branches of chemistry, it is necessary to convert one functional group to another. This is a very common process or reaction, known as Functional Group Interconversion (FGI). This method or reaction is used in the pharmaceutical and chemical industries and in perfume manufacturing.
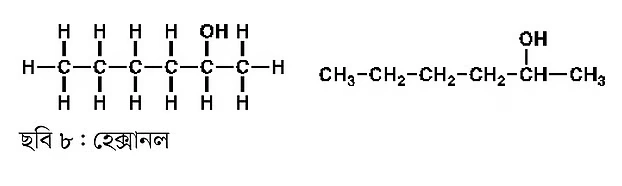
The compound below (Figure 8) contains six carbon atoms, so its name is hexanol. From hexane to hexanol.
To draw 2-hexanol, we will not use any of the previous methods shown above. Instead, we will use the modern drawing method (Figure 9).
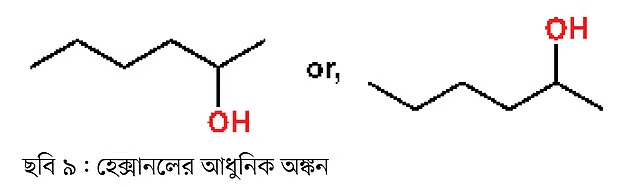
Such alcohols are called branched or internal alcohols, since the alcohol group is not attached to a terminal carbon atom. In contrast, the compound below is 1-hexanol (Figure 10), where the alcohol group is attached to the terminal carbon. This type of compound is called terminal or linear alcohol.
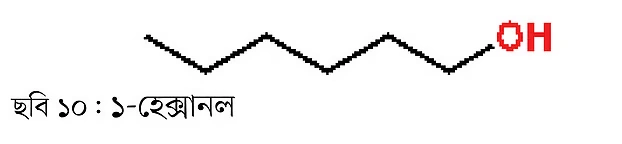
So, 1-hexanol is a linear alcohol and 2-hexanol is a branched alcohol. If you notice, these two compounds have the same molecular mass. However, they are different chemical compounds because the functional group (alcohol group) is attached to different carbon atoms. Therefore, they are each other’s isomers. This type of isomer is called a regioisomer. In other words, they can also be called positional isomers. This is a very important concept in chemistry. Regioisomers are distinct compounds with different physical and chemical properties.
Similarly, other functional groups can replace the alcohol group to form different compounds. For example, instead of the alcohol group, we can use halogen atoms (F, Cl, Br, I). Like 1-hexanol, we can draw 1-bromohexane by replacing the alcohol group with a bromine atom, resulting in a different compound.

Like alcohols, this compound can also be linear or branched, depending on which carbon the bromine atom is attached to.
These types of compounds can be cyclic as well, like bromo-cyclohexane. In the previous part, I discussed a stable conformation of cyclohexane—the chair conformation.
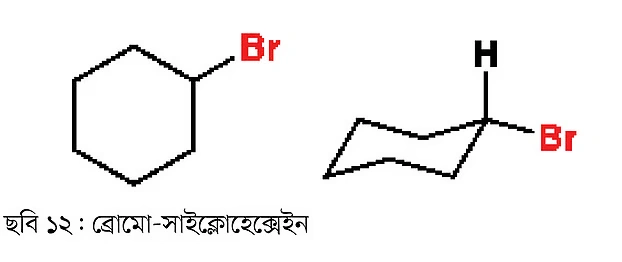
The two compounds above are actually the same compound, just presented differently. The reason for drawing with the chair conformation is to familiarize you with it. You should practice drawing any cyclohexane compound this way, as it has particular importance. There will be a more detailed discussion about this in future installments.
Learning the modern methods for drawing chemical structures will make understanding and explaining many things much easier, including chemical reactions. Most students in Bangladesh don’t understand chemical reactions well or find them difficult, and one reason for this is their lack of understanding of chemical structures. In the next part, we will cover more fascinating topics.
References:
This writing is adapted from an article published in Bigganchinta Magazine.
Author: Dr. Rouful Alam, Researcher, University of Pennsylvania, USA.











Leave a comment