What is a lead-acid battery?
Battery Basics
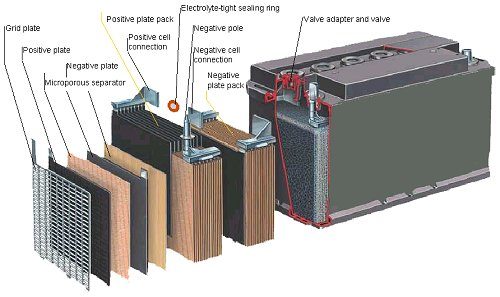
A lead-acid battery is made from plates, lead, and lead oxide [along with some other components for density, strength, etc.] mixed with 35% sulfuric acid and 65% water. This acid-water mixture is called the electrolyte, which produces electrons through chemical reaction. The sulfuric acid concentration in the electrolyte can be measured with a hydrometer for battery testing.

Battery Types

Generally, there are two types of batteries,
Deep cycle: These can withstand many charge and discharge cycles. This type of battery is also called a marine battery. Their specifications are usually written like this: 12V7AH battery, meaning it is 12 volts and is capable of supplying 7 amperes of current in one hour or running at 7 amperes for 1 hour. They contain thick plates that can retain charge for a long time and discharge slowly.
Starting battery or cranking battery: These are primarily for vehicles. Vehicles or engines have a powerful starting motor (called a self) which only needs to run for 15–30 seconds to start the engine, meaning a short burst of 25–150 amperes is needed to run the self. After this, the battery is not much in use for power, as it gets recharged again from the engine’s alternator. These batteries have thin plates that can produce a high current or amperage.
Deep cycle batteries are used in UPS/IPS, and starting batteries cannot provide good service in UPS/IPS applications.
mamun2aatgmail









Leave a comment