Guest Author: Rauful Alam
Writer and Researcher
Email: [email protected]
The modern method of drawing organic compounds has been discussed in detail over the previous three installments. I hope that teachers and students will practice the modern style. In the following episodes, there will be extensive discussions about reactions in organic chemistry.
A major portion of chemical reactions deals with organic chemistry. Many students simply memorize chemical reactions by rote. In fact, chemical reactions should be explained with the help of certain logics, facts, and theories. Various theories have been applied in different ways to explain chemical reactions. Many theories have been established through experiments—we call this experimental validation. In addition, many theories have been established with the help of quantum chemistry.
In this part, we will discuss the chemical bonding and other properties of certain compounds. We use several theories to explain chemical bonding. One of the most basic is the ‘Lewis structure’. However, you can’t explain everything in detail with Lewis structure alone. That’s why other theories, such as VBT or Valence Bond Theory, VSEPR or Valence Shell Electron Pair Repulsion Theory, and Molecular Orbital Theory (MOT) are used. The last two are primarily for advanced levels. My goal with these installments is to help students overcome their fear of chemical reactions. So, I will try to explain core concepts as simply as possible, with only limited discussion of advanced theories. First, let’s drive away the fear, then go deeper. Many fundamental concepts can be explained without advanced theory.
From the group number of any element, we can tell how many electrons are in its outer shell. It’s a very simple matter. No need to memorize it tediously; you don’t even have to remember it. Just by looking at the periodic table, you can say it.
It is best to keep a periodic table nearby while reading these segments. The periodic table contains a lot of information. You just need to know how to extract and use the data from there, and how to remember the facts from the periodic table.
Your school textbook has surely discussed the Lewis structure. The atomic number of carbon is 6, so there are 6 protons in its nucleus. The atom has a neutral charge, meaning there is no net charge. Thus, it also has 6 electrons. If there are electrons, next comes the arrangement—how many electrons are in which orbit or shell. The first orbit holds a maximum of two electrons. So, carbon’s second or outermost shell contains 4 electrons. Even if you don’t remember this, you can figure it out by looking at the periodic table.
Carbon is an element in group 14 of the periodic table. The old name for group 14 was group IVA. That’s because the transition metals are present in the 10 groups from group 3 to 12. Most organic chemistry reactions can be explained without considering these metals. So if you exclude the 10 transition metal groups from carbon’s group number 14, you get group 4 (14-10), which actually refers to the old IVA group.
From the group number of any element, we can tell how many electrons are in its outer shell. It’s a very simple matter. No need for tedious memorization; no need to remember it separately. Just a glance at the periodic table reveals the answer. For example, sodium is in group 1—meaning it has 1 electron in its outermost shell. The same is true for lithium and potassium. Nitrogen’s outer shell contains 5 electrons because nitrogen is a group 5 (15-10) or VA element. Carbon’s outer shell contains 4 electrons because carbon is a group 4 (14-10) or IVA element. Why is the number of outer shell electrons important? Because chemical reactions mean the exchange of electrons—and this exchange happens with the outermost electrons. So, knowing the number of electrons in the outermost orbit (shell) is important.
Carbon is an element in group 14 of the periodic table. The old name for group 14 was group IVA. That’s because the transition metals are present in the 10 groups from group 3 to 12. Most organic chemistry reactions can be explained without considering these metals.
The carbon atom has four electrons in its outermost shell or orbit. So, how many more electrons can a carbon atom accommodate? Four more. Why? Because with 4 additional electrons, carbon’s outer shell would have 8 electrons. Then, it could achieve a stable electron configuration like neon. This tendency to fill up to 8 electrons is called the octet rule. Using the octet rule, you can easily draw the Lewis structures of methane and ethane. The Lewis structure here illustrates covalent bonding.
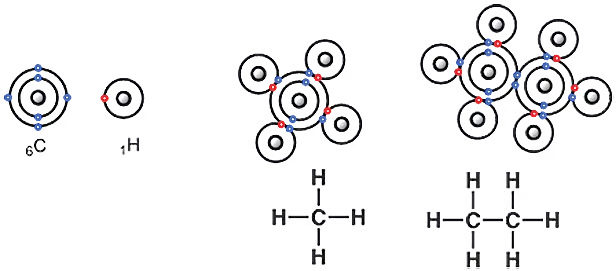
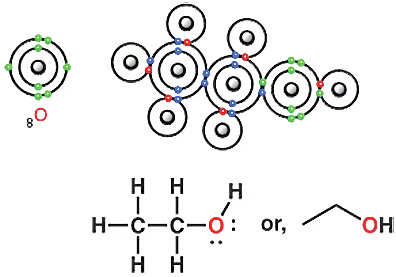
Once you know the structure of ethane, you can also draw the structure of ethanol or ethyl alcohol. Ethanol is a very important compound. Since this molecule contains an OH group, it is called an alcohol.
The oxygen atom in ethanol also forms bonds with carbon and hydrogen following the octet rule.
Do we see any electrons in carbon that do not take part in bonding? No, we do not. But we do see this in the case of oxygen. Four electrons of oxygen (shown in green) do not participate in chemical bonding. Two pairs of electrons do not participate in bonding, so these are called lone pairs or non-bonding electron pairs.
In the case of oxygen, you see two such pairs. Since ethanol’s oxygen contains two lone pairs—that is, two pairs of non-bonded electrons—this applies to all alcohol compounds. For methanol, isopropanol, or any other alcohol, we can apply the same principle.
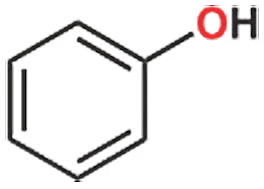
If the alcohol group is attached to benzene, it is called phenol, and in such compounds, the oxygen atom also contains two lone pairs. This is an important fact to remember.
In the periodic table, the element just below oxygen is sulfur. Both are in the same group. Thus, they share many properties. When sulfur replaces oxygen, the compound’s name includes ‘thio’ or ‘thiol’—for example, ethanol becomes ethanethiol, and phenol becomes thiophenol.
The carbon-sulfur bond is larger than the carbon-oxygen bond. This, too, can be understood from the periodic table. Oxygen is a second-period element—so oxygen has two orbits. Sulfur is a third-period element.
Whether it’s ethanethiol or thiophenol, the sulfur atom still contains two lone pairs. The straightforward logic is: oxygen and sulfur are both in the same group. So, if the alcohol’s oxygen has two lone pairs, the analogous sulfur compound’s sulfur will also have two. This is the logic to remember here. I don’t even have to consider sulfur’s electron arrangement separately.
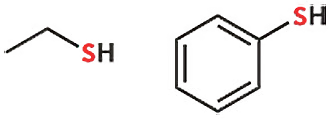
Are the carbon-oxygen bond length in ethanol and the carbon-sulfur bond length in ethanethiol the same? Bond length is basically the distance between the nuclei of the two atoms. There is no need to memorize bond lengths—just understanding the idea is enough. The carbon-sulfur bond is larger than the carbon-oxygen bond. This also can be understood from the periodic table. Oxygen is a second-period element, so oxygen has two orbits. Sulfur is a third-period element, so it has three orbits. More orbits mean a larger atomic size. A larger atom results in a longer chemical bond. Longer chemical bonds are weaker, meaning they require less energy to break. These facts are important to remember as well.
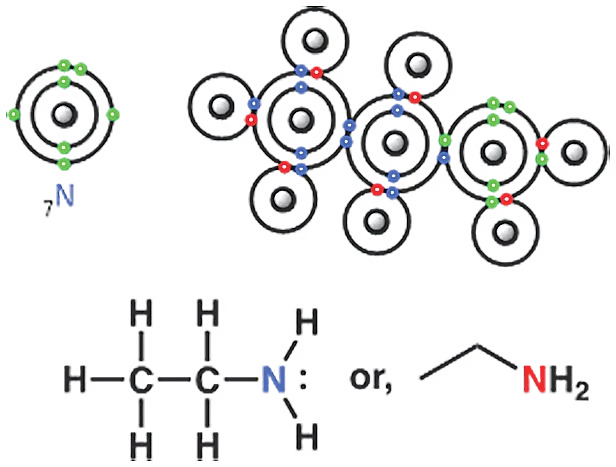
Now let’s move on to nitrogen compounds. The nitrogen analogue of ethanol is called ethyl amine. Here, nitrogen replaces ethanol’s oxygen. From the Lewis structure, you can tell that ethyl amine’s nitrogen does not have two lone pairs; it has only one lone pair—that is, one pair of electrons not involved in bonding.
The nitrogen in amine compounds has one pair of electrons, i.e., one lone pair. The first compound is ethyl amine, the second is diisopropylethylamine (DIPEA), the third is triethylamine (TEA), and the last one is aniline.
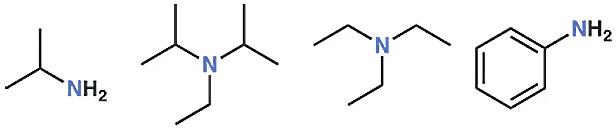
Many students may ask, why does nitrogen form bonds with two hydrogens, and why is there only one lone pair? The answer lies in the number of outer shell electrons in nitrogen. Nitrogen has 5 electrons in its outermost shell. So, it needs just 3 more electrons to complete its octet. Thus, a nitrogen atom can take a total of 3 electrons from two hydrogens and one carbon—making three bonds (shown in different colors). So not all 5 of nitrogen’s electrons can form bonds. Otherwise, the outer orbit would have 10 electrons, which does not fit the octet rule. So it makes three bonds, while the remaining two electrons (one pair) do not participate in bonding. By using this simple and easy logic, the other compounds can also be explained.
So, the nitrogen in amine compounds has one pair of electrons or one lone pair. The first compound is ethyl amine, the second is diisopropylethylamine (DIPEA), the third is triethylamine (TEA), and the last is aniline. These are different amines. But in every compound, the nitrogen has a single lone pair. There is very little difference in the bond length between a carbon-nitrogen single bond and a carbon-oxygen single bond. Both are nearly the same in length and energy.
I specifically discussed the lone pairs of oxygen, sulfur, and nitrogen. That’s because these compounds will frequently come up as examples.
I will end this section with a special type of compound. In the above examples, the oxygen, sulfur, and nitrogen atoms contained a lone pair. In this example, however, you will not find a lone pair—rather, you will find a vacancy!
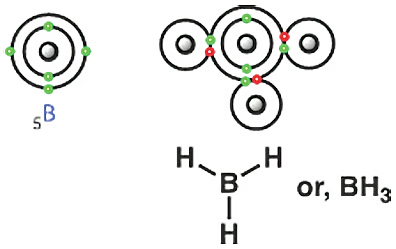
The following compound is called borane. Boron is a group 13 element. That means it is also a group (13-10) = 3 or IIIA element. So, boron has three electrons in its outermost shell. Therefore, boron can form bonds with three hydrogens and acquire a total of 6 electrons. According to the octet rule, boron still needs two more electrons to reach 8. So, there are no lone pairs or non-bonding electrons on boron; instead, there’s a vacancy. Boron is eager or ready to accept a lone pair or an extra pair of electrons.
If all 5 of nitrogen’s electrons formed bonds, the outer orbit would have 10 electrons, not following the octet rule. That’s why only 3 electrons participate in bonding and the remaining 2, i.e., one pair, do not.
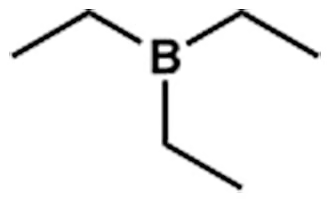
Just like triethylamine (TEA), there is also a compound called triethylborane (TEB). The difference is, in amines, the nitrogen has a lone pair. In borane, there is none. In fact, it can accept a lone pair.
Boron (B) is eager to accept a pair of electrons or a lone pair. Carbon has no lone pair (see examples of methane, ethane). Nitrogen has one, oxygen has two, and fluorine has three.
B, C, N, O, F—these elements are present consecutively in the periodic table. If you think of them with numbers, their lone pairs can be counted as 1, 0, 1, 2, 3. What an amazing fact! Have you ever thought of it this way?
References:
This piece was adapted from an article published in Bigganchinta Magazine.
Author: Dr. Rauful Alam, Researcher, University of Pennsylvania, USA.











Leave a comment