In 1974, a young professor at the University of Melbourne, Australia, Richard Robson, was building models using wooden balls to help students visualize molecular structure. Each ball represented an atom, and each wooden rod symbolized a chemical bond. He understood that if the holes in these balls were drilled randomly, the model would be meaningless, because each atom bonds in specific numbers and directions. Carbon bonds in four directions, nitrogen in three, and chlorine in only one. So, the placement of holes in each ball depended on its “chemical nature.”
When he received the drilled balls from the workshop and started assembling molecular structures, Robson suddenly noticed a profound truth: the position of holes on each atom served as a kind of “instruction.” Hidden within the geometry of these holes were the rules of bonding—where, at what angle, and how far another atom could fit. This sparked a fundamental idea in his mind: if we could use the real geometric tendencies and electronic attractions of atoms and molecules to guide their assembly, could they spontaneously form complex, orderly structures on their own?
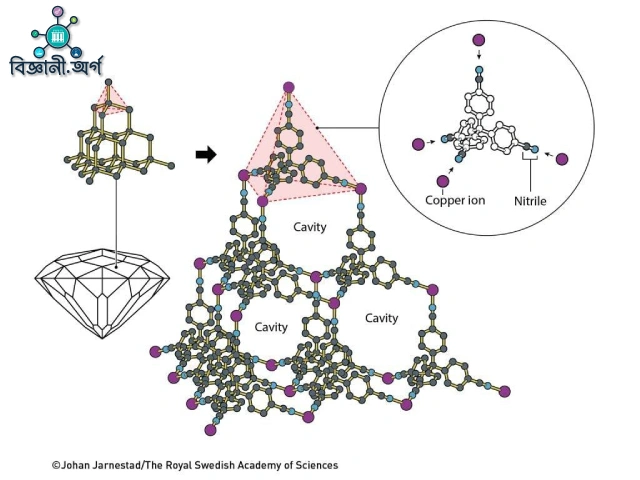
To turn this idea into reality, Robson looked to a perfect example from nature—diamond. In diamond, each carbon is tetrahedrally bonded to four other carbons, forming an infinite three-dimensional lattice (see image). If, instead of carbon, a metal ion with a preference for tetrahedral geometry could be used, a similar lattice might be built. He chose the positively charged copper ion (Cu(I)), whose (3d¹⁰) electron configuration gives it stability and a preference for being surrounded by four ligands. Next, he needed a molecule capable of extending four arms in equal directions (like carbon does) with, at each arm’s end, a group capable of bonding to the copper ion. That ligand was 4,4′,4″,4‴-tetracyanotetraphenylmethane, a sturdy, stable molecule with a central carbon bearing four phenyl rings, each terminated with a nitrile (–C≡N) group (see image). The lone pair electrons on the nitrile’s nitrogen could readily donate to the empty 4s/4p orbitals of Cu(I), forming strong coordination bonds.
This concept materialized in 1989, when Robson and his colleague Bruce Hoskins published a landmark study in the Journal of the American Chemical Society (comment). For the first time, they showed that by using carefully selected metal ions and geometrically precise organic ligands, one could intentionally create an infinite, three-dimensional, crystalline lattice (see image). The synthesis started with tetraphenylmethane, which was brominated using bromine and iron catalyst to yield tetrabromotetraphenylmethane. This compound was then refluxed with CuCN in DMF, replacing all four bromines with –CN groups to yield pure 4,4′,4″,4‴-tetracyanotetraphenylmethane, the ligand. Reacting this ligand with Cu(CH₃CN)₄BF₄ in a nitrobenzene/acetonitrile mixture, and slowly evaporating the solvents, they obtained colorless crystals—whose structure was ([Cu(C(C₆H₄CN)₄)]BF₄·xC₆H₅NO₂) (x ≈ 8.8). The IR spectrum showed a strong ν(C≡N) band at 2240 cm⁻¹ and a ν(BF₄⁻) band at 1060 cm⁻¹—clear evidence of Cu-N coordination.
When they performed single-crystal X-ray diffraction, a remarkable picture emerged. Each Cu(I) ion was surrounded tetrahedrally by four nitrile nitrogens (Cu–N distance 2.03 Å), and each ligand used its four arms to connect four different copper centers (see image). The resulting structure was topologically a diamond-like “diamondoid” lattice, with both types of nodes (Cu and central carbon) maintaining tetrahedral geometry (see image). Each lattice unit was about 8.86 Å across, with vast cavities of around 700 ų running through the structure, able to house solvent and anions. About two-thirds of the crystal’s volume was occupied by liquid nitrobenzene and mobile BF₄⁻ counterions, but the lattice itself was highly crystalline—porous yet ordered.
One striking feature of these crystals was their “responsive stability”—meaning it was possible to exchange the internal anions while leaving the lattice intact. They showed that soaking the crystal in an NBu₄PF₆/nitrobenzene solution replaced the BF₄⁻ anion with PF₆⁻, evidenced in IR by new bands at 830 and 560 cm⁻¹. Yet, the crystal’s external shape remained unchanged. This indicated that the Cu-N coordination network was strongly maintained, while ion exchange happened within the lattice’s cavities.
Chemically, the lattice’s bonds were essentially σ in character—the lone pair on nitrogen donating to the empty 4s/4p orbitals of Cu(I). This created an “electronically saturated” σ-network—solid, yet electronically stable. The rigid geometry of the tetranitrile ligand propagated local tetrahedral bonding into a regular three-dimensional lattice over long distances.
This work marked a fundamental turning point in the history of chemistry. It demonstrated that coordination chemistry, previously limited mainly to discrete complexes, could be used to build infinite, three-dimensional, crystalline, porous frameworks. This laid the foundation for the modern concept of metal-organic frameworks (MOFs)—materials designed like an architect’s bricks and beams, joining metals and organic ligands according to targeted topology and functionality.
However, those early compounds were quite fragile, easily breaking apart and losing stability upon exposure to air or solvents. Many thought these structures were merely fascinating laboratory curiosities, with no real-world practical value. Yet, some visionary chemists sensed that Robson’s work held the key to a new horizon in chemistry’s future.
It was in this continuation that two researchers emerged: Susumu Kitagawa and Omar Yaghi. They gave firm foundation to Robson’s theoretical idea, developing advanced synthesis and precision design to create stable, controllable structures that were truly porous coordination frameworks. They demonstrated how metal ions and organic ligands could be assembled so that the resulting lattice was both robust and, on the inside, hollow—open for guest molecules, gases, or ions, forming a new architecture in chemistry.
Decades later, this very concept has been recognized with science’s highest honor. Richard Robson, Susumu Kitagawa, and Omar Yaghi were all jointly awarded the Nobel Prize in Chemistry in 2025, for their architecture of molecules—an achievement that began with simple classroom wooden balls and culminated in infinite, ordered crystalline networks where molecules themselves became the architects.
Thus, Robson’s story begins with a classroom toy and ends in an entirely new philosophy of chemistry.
Sources and Photo: Nobel Prize in Chemistry 2025; J. Am. Chem. Soc. 1989, 111(15), 5962–5964
Md Yeasin Pabel
Graduate Student
Department of Chemistry
University of Florida










Leave a comment