Guest Author: Rauful Alam
Writer and Researcher
Email: [email protected]
The previous part discussed the drawing of structures of polybond and alcohol-based compounds. In this part, we will discuss a few more functional group-containing compounds. If you have read the first two parts, this discussion will be easier to understand.
Polybonded Compounds
If an organic compound contains only carbon-carbon single bonds (C-C bond), they are called saturated compounds. If there is a double (C=C bond) or triple bond between two carbons, then these are called unsaturated compounds. One point to remember: if a compound contains carbon-oxygen double bonds, or carbon-nitrogen double or triple bonds, these are NOT considered unsaturated compounds. Whether a compound is saturated or unsaturated depends only on the carbon-carbon bonds.

Alcohols have a carbon-oxygen single bond, while aldehydes have a carbon-oxygen double bond. That’s why the names of aldehydes end with “nal”.
Similarly, ketones can also be made without changing the carbon chain—like in the examples below where 2-hexanol and 2-hexanone are shown. As a reminder, 1-hexanol and 2-hexanol are both alcohols and both have the same number of carbon atoms, but they are different compounds, with different physical and chemical properties. The only difference is that the alcohol group is attached to a different carbon. These two compounds are regioisomers of each other.
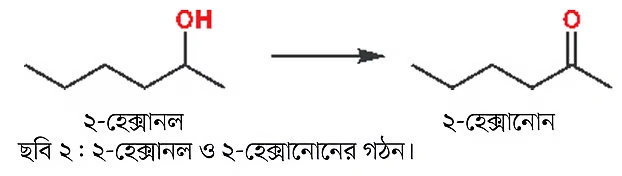
On the other hand, 1-hexanal and 2-hexanone are both carbonyl compounds, but the first is an aldehyde, and the second is a ketone. In ketones, the carbonyl carbon atom is attached to two other carbons.
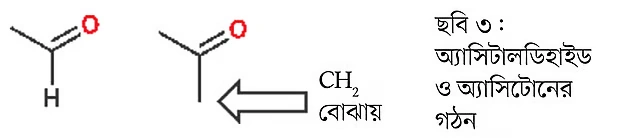
The simplest ketone is acetone. Above, the structures of acetaldehyde and acetone are shown (Image 3). Acetone is a volatile liquid, colorless, and commonly used as a solvent. Acetone is used as a nail polish remover. During World War I, British soldiers made explosives from acetone.
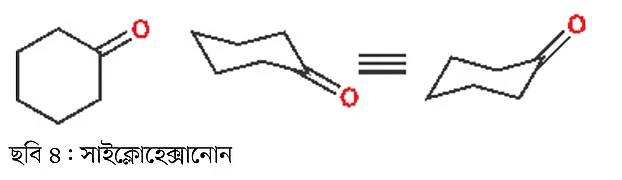
During the discussion on cyclohexene, we also looked at its chair conformation. Likewise, drawing the cyclohexanone molecule in different ways can help in discussion and understanding. All the drawings in the image below are correct (Image 4). However, drawing it in chair conformation is recommended for practice.
When an oxygen atom is attached to two carbon atoms, the compound is called an ether. Basically, if the oxygen atom of an alcohol group is attached to a carbon instead of hydrogen, it’s an ether (Image 5).
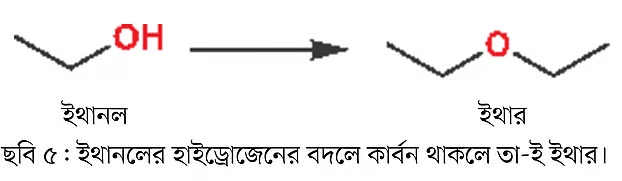
For example, if you replace the hydrogen atom bonded to the oxygen in ethanol with another ethyl group, you get diethyl ether, or simply ether. Ether was once used as an anesthetic. It is still widely used as a solvent.
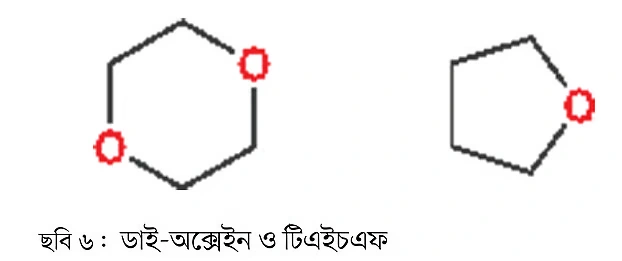
Ethers can also be cyclic. The hexagon-shaped compound above is called dioxane, and the pentagon-shaped one is tetrahydrofuran or simply THF (Image 6). Both are extensively used as solvents. In later installments, these compounds will be used as examples, so I am introducing them now.
At this stage, let’s look at a few examples of carboxylic acid compounds. We’ll also discuss some compounds derived from the carboxyl group. Surely you have heard of fatty acids—generally, organic acids with carbon chains are called fatty acids.
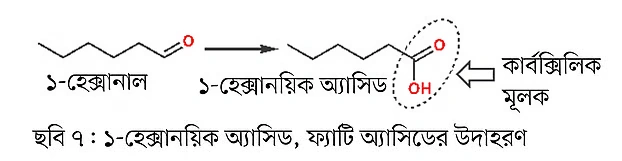
For example, 1-hexanal is an aldehyde compound. If the carbonyl carbon of that compound is attached to an OH group, it becomes a carboxylic acid group—in other words, a carboxylic acid or 1-hexanoic acid. Fatty acids can also contain carbon-carbon double bonds, meaning they can be unsaturated fatty acids. The above fatty acid example is of a saturated fatty acid. The edible oils we consume contain both saturated and unsaturated fatty acids, so those oils have carbon-carbon double bonds. And the presence of double bonds means cis or trans isomers can exist. The previous part discussed this topic.
If a fatty acid or edible oil contains trans-double bonds, it is called trans fat, which is harmful to the body. Doctors recommend that we avoid trans fat because our bodies cannot easily break down trans-double bonds. Vegetable oils do not contain trans fats—they contain cis-double bonded fatty acids.
Two other important groups, esters and amides, also arise from carboxylic acids. The sweet smell of fruits is mostly due to ester compounds. For instance, the first molecule is acetic acid, the main acid in vinegar. If you replace the hydrogen of the hydroxyl group in acetic acid with an ethyl group, you get an ester—ethyl acetate. Ester compounds are also widely used as solvents.
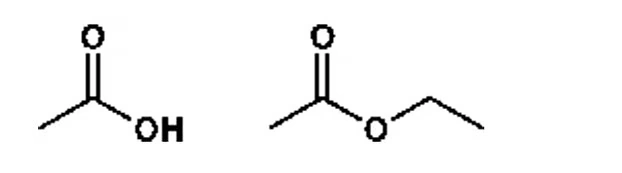
Image 8
Following modern drawing methods, these compounds can now be drawn very quickly and easily. By increasing the length of the carbon chain, many compounds can be rapidly illustrated. Below is another example of an ester. In fact, by replacing the ethyl group of ethyl acetate with an isoamyl group (five carbons), you can draw this ester. The distinctive smell of bananas comes from this ester compound (Image 9).

The sources of taste and aroma are mostly organic compounds. These molecules prominently feature alcohol, aldehyde, ketone, and ester functional groups.
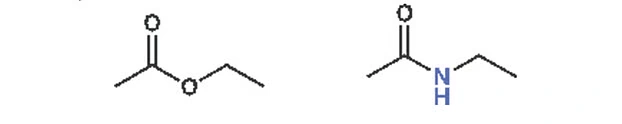
Image 10
If you replace the oxygen of an ester with a nitrogen atom, you get an amide. Above, examples of both ethyl acetate and amide are shown. Just below, you can see formic acid along with dimethylformamide.
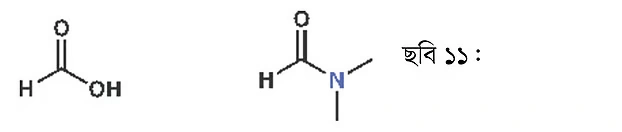
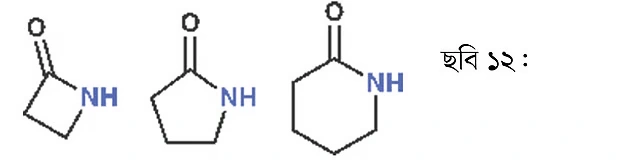
Dimethylformamide (DMF) is used as a solvent. As this solvent has a high boiling point (about 150°C), it is used in reactions at high temperature. Amide group-containing compounds are also found in drugs. For instance, a key part of the penicillin molecule is a cyclic amide. This type of cyclic amide is called a lactam. If it’s a four-membered ring, it’s called a beta-lactam. In a five-membered ring, it’s a gamma-lactam, and in a six-membered ring, a delta-lactam. Penicillin contains a beta-lactam ring.
Now let’s discuss functional groups containing nitrogen atoms. Although amides contain nitrogen, amides are considered derivatives of carboxylic acids. Among nitrogen or amine group-containing compounds, ammonia is the smallest. Replacing the oxygen atom in a water molecule with nitrogen gives ammonia. Conceptually, this is a way to draw analogy. Amines are alkaline compounds.
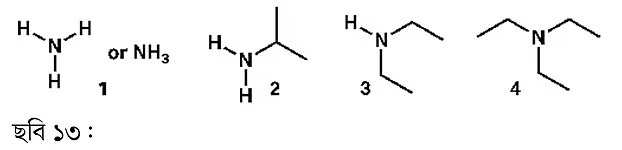
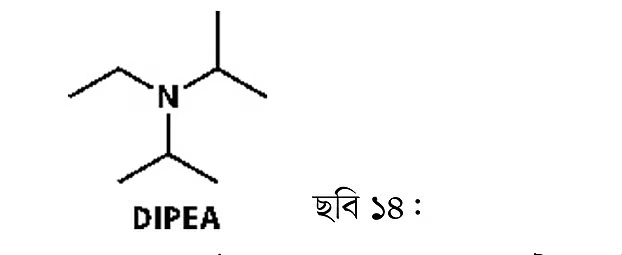
If the hydrogens in ammonia are replaced by other groups, different amines are formed. For example, adding an isopropyl group makes (2) isopropylamine. Adding two ethyl groups gives (3), diethylamine. Diethylamine can be compared to ether: ether contains oxygen, while diethylamine contains nitrogen. Since ammonia has three hydrogens, all three can be replaced by carbon chains. For instance, adding three ethyl groups produces triethylamine (4). Triethylamine (TEA) is widely used as an organic base. Another widely used base is diisopropylethylamine (DIPEA), also known as Hunig’s base. Its uses will be discussed in the future, so it’s recommended to read and practice repeatedly to remember these.
Now, let’s discuss the drawing of aromatic compounds. Since aromatic compounds belong to a special class, they have not been discussed so far.
Cyclohexene is a cyclic compound, as discussed in the second part. This compound has one double bond and six carbon atoms. If a similar compound has three double bonds, it becomes benzene—a completely different type of molecule.
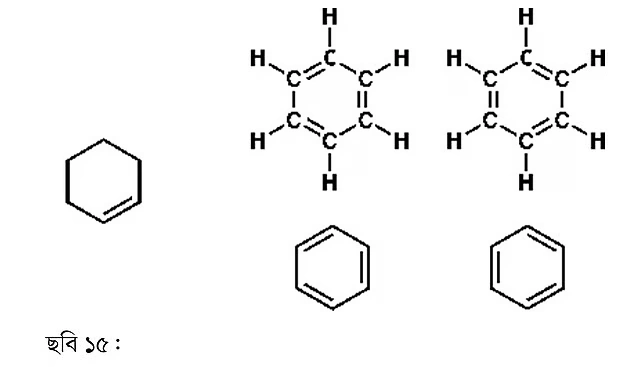
We must practice drawing benzene according to modern conventions. There’s no need to show every carbon-hydrogen bond. Some people draw a circle instead of showing all three double bonds; such representations are found in various books, though this practice is going out of style. The latest books show all three bonds separately, as it’s more useful for explaining chemical reaction mechanisms.
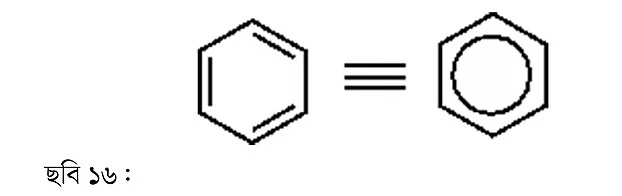
Benzene is a completely planar molecule. It is both a cyclic and aromatic compound—a completely separate class of molecule. By attaching various functional groups or aliphatic chains to the benzene ring, many compounds can be drawn. When a nitrogen group is attached to a benzene ring, those are called hetero-aromatic compounds.
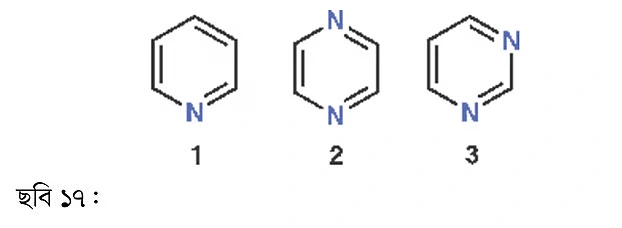
The first molecule is pyridine—an aromatic liquid, used as an organic base. The second compound is pyrazine, which has a nutty aroma but isn’t a liquid like pyridine; rather, it is solid at room temperature. The third is pyrimidine. Both the second and third compounds contain two nitrogen atoms. The drugs we take most often contain hetero-aromatic rings in their molecules.
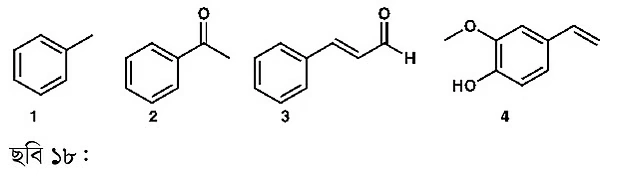
Above are examples of a few more aromatic compounds. When a methyl group is attached to the benzene ring, it’s called toluene (1). Toluene is used as a solvent. The second compound is acetophenone—a ketone group attached to benzene. This compound is found in almonds. The third compound is my favorite: cinnamaldehyde, which gives cinnamon its characteristic aroma. The last compound has a very strong odor which I do not particularly like; this is eugenol (4), responsible for the aroma of cloves. Every compound mentioned here, I’ve used in the lab. Organic chemists routinely handle countless such molecules.
Let’s finish this part with a few examples of drug molecules. The first compound below is paracetamol—a drug nearly a century old. Here you can see both an amide group and an alcohol group attached to the benzene ring. The second is aspirin, also nearly a century old.
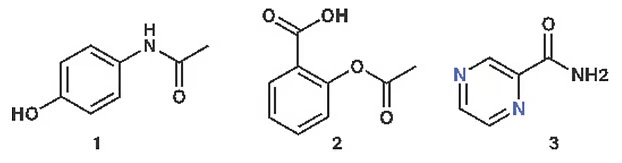
Image 19
The aspirin molecule contains both carboxyl and ester groups. The last molecule is a drug used for tuberculosis. This molecule contains a pyrazine ring and an amide group.
With these three parts, we’ve concluded the modern rules for drawing organic molecules. The next installments will discuss simple reaction mechanisms. Examples like these, or similar ones, will appear in those discussions. There will also be contextual discussion on chemical bonds. To easily understand reactions, it is important to be familiar with these types of illustrations by practicing. I hope school and college teachers will use this illustration style when teaching students.
Reference:
This article is adapted from a feature published in the Bigganchinta Magazine.
Author: Dr. Rauful Alam, researcher, University of Pennsylvania, USA.










Leave a comment