Guest Author: Rauful Alam
Writer and Researcher
Email: [email protected]
🔗Easy Lessons on Chemical Reactions
To understand and explain chemical reactions, especially in organic chemistry, you need a solid grasp of the following concepts.
1. Electronegativity; 2. Acidity and basicity, which we denote with pKa; 3. Conjugate acids and conjugate bases; 4. Electron-donating groups and electron-withdrawing groups; 5. Electrophiles; 6. Nucleophiles; 7. Bond length and bond strength; and 8. Sterics.
By applying or analyzing these aspects in any chemical reaction, you can gain a good understanding of the process. As I mentioned before, there is nothing to memorize about reaction mechanisms; instead, we use logic to figure them out or understand them. For completely new reactions, sometimes a lot of experiments are performed to establish the mechanism. Anyway, let’s discuss the points mentioned above first.
01. Electronegativity: In Bangla, this is called “todiot rinatomkota.” In a covalent bond, when the two atoms are different, there is a tendency for atoms to attract or hold onto the bonding electrons towards their own nucleus. The atom with higher electronegativity pulls the bonding electron more toward its own nucleus. The more protons in the nucleus and the smaller the size of the atom (or the fewer orbits it has), the higher its electronegativity.
The values of electronegativity have been determined, and there’s no credit in memorizing them. However, it’s important to have an idea of which atoms have higher or lower electronegativity. This understanding comes from the periodic table itself.
Electronegativity increases as you move from left to right across a period in the periodic table. But as you go from top to bottom within a group, electronegativity decreases. The first element in the second period is lithium. Moving right from lithium, you get beryllium, boron, carbon, nitrogen, oxygen, and fluorine. Since noble gases do not form covalent bonds, their electronegativity is not considered. So, carbon’s electronegativity is higher than boron’s, and nitrogen’s, oxygen’s, and fluorine’s are higher than carbon’s. In fact, fluorine has the highest electronegativity.
02. Acidity and Basicity: To understand the acidity and basicity of functional groups in organic compounds, we use pKa. An easy way to remember is: when pKa is low, the substance is more acidic or a stronger acid. When pKa is high, it’s less acidic or a weaker acid. We have determined the pKa values for many compounds and there are lists available. So you don’t need to memorize pKa values; instead, familiarize yourself through practice.
For example, acetic acid (1) has a pKa value close to 5. That’s an acid; meaning, if a compound has a carboxylic group, its pKa will be near 5. However, if there is an electron-withdrawing group (EWG) directly attached to the carboxylic group, the acidity increases substantially. A higher acidity means an even lower pKa value. For instance, trifluoroacetic acid (TFA, 2) has a pKa value close to zero. pKa can even be negative.
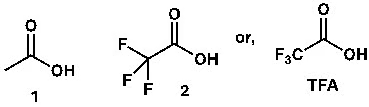
Figure 1: Trifluoroacetic Acid
Phenol has a pKa of about 10. So between phenol and acetic acid, which is the stronger acid? Clearly, acetic acid. And alcohols, like ethanol, have a pKa around 15; meaning, ethanol is a very weak acid.
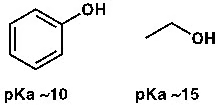
Figure: A higher pKa means a weaker acid.
You may wonder why it’s important to know about pKa values. Because if an acid is strong—that is, has a low pKa—then you can use a weak base to remove its proton. This is called deprotonation. The pKa values of many bases have also been determined, though by a base’s pKa we mean the pKa of its conjugate acid. For example, triethylamine’s conjugate acid has a pKa of around 10.
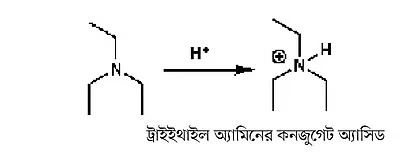
Figure 2: The pKa of a base refers to the pKa of its conjugate acid. Here it shows the pKa of triethylamine’s conjugate acid.
When you understand the pKa values of acids and bases, you can easily decide which base to use with which acid. Suppose you need to deprotonate phenol—that is, remove its acidic hydrogen. You’ll need a base. Which one should you choose?
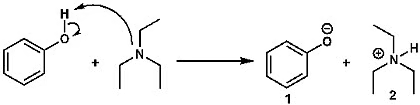
Figure 3: Deprotonation of Phenol
Think about the reaction above. Phenol is a weak acid. Why? Because its pKa is about 10. So, to generate the phenoxide ion (1) by deprotonating phenol, suppose we use triethylamine (TEA). Will this work? To answer, we must know the pKa of the conjugate acid of triethylamine, which is also around 10. Since both compounds have about the same pKa, triethylamine will be able to deprotonate phenol, but the process will be slow. The reaction will maintain equilibrium. (Triethylamine has been discussed previously, and the equilibrium of reactions will require further discussion elsewhere.)
But what if we consider acetic acid and triethylamine? Acetic acid has a pKa of about 5 and triethylamine’s (conjugate acid) pKa is about 10; meaning, in this case, the base is much stronger. As a result, acetic acid and triethylamine will react rapidly, producing the acetate ion (1) that does not revert to acetic acid—so this reaction’s equilibrium is essentially one-way.
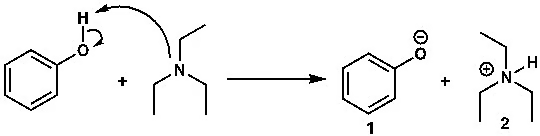
Figure 4: Acetic acid and triethylamine react rapidly, producing acetate ion (1). This reaction is one-way.
You can imagine how fast the reaction between trifluoroacetic acid and triethylamine would be. Consider their pKa values and think about it yourself.
Now, let’s discuss conjugate acids and conjugate bases. The two preceding examples make this easy to understand. When the acidic hydrogen (also called a proton) is removed from an acid by deprotonation, the negatively charged ion formed is the conjugate base of that acid. So, in the above reaction, acetate (1) is formed from acetic acid and is its conjugate base.
Why do we call it a “conjugate”? Because if you remove a proton from acetic acid, you get acetate (1); and if acetate accepts a proton, it becomes acetic acid again. They are a pair, connected to each other.
Similarly, the conjugate acid of triethylamine is the triethylammonium ion (2). Since the ion has a proton, we call it an acid now; but it comes from a base.
In simple terms, an acid gives rise to its conjugate base, and a base gives rise to its conjugate acid.
If an acid is strong, its conjugate base will be weak. What does this mean? For example, in the case above, acetic acid is relatively strong. Thus, its conjugate base, the acetate ion, is a weak base. For this reason, the acetate ion cannot snatch a proton from triethylammonium amine.
So is phenoxide a weaker or stronger conjugate base than acetate? That’s for you to think about.
References:
This article was compiled from a published essay in BigganChinta Magazine.
Author: Dr. Rauful Alam, Researcher, University of Pennsylvania, USA.











Leave a comment