The most important driving force behind the advancement of human civilization is undoubtedly electricity. The journey began with coal, followed by petrol and diesel. But Earth’s resources are not infinite, so humanity continues to search for new sources of energy—to keep this civilization moving forward. Among all the sources of energy on Earth, ultimately we come back to the Sun. Sunlight is the main source of energy for all life on Earth. One of the greatest dreams of modern science is to directly convert sunlight into electricity and make it easily accessible to everyone. This dream is called solar energy, and the device to capture this power is called a solar cell.
Limitations of Silicon Solar Cells
The solar panels we often see on rooftops are mostly made using silicon. These silicon solar cells have long been a reliable technology, but they come with significant drawbacks.
First, manufacturing silicon solar cells requires very high temperatures and expensive equipment. Second, they are relatively heavy and prone to breakage. But the biggest issue is that their cost still remains beyond the reach of ordinary people in many countries.
That’s why scientists began to wonder—could there be a new material that’s cheap, easy to make, and can efficiently convert sunlight into electricity?
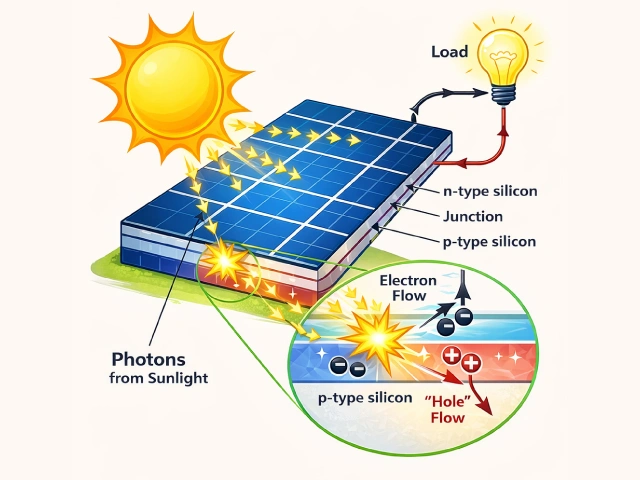
How energy is generated from a solar panel: When sunlight hits the panel,
special materials inside create electricity.
At this moment, tiny particles (electrons) start moving, and that’s where the electricity comes from.
This electricity can be used to power lights, fans, or charge a mobile through wires.
In this way, solar panels provide clean and environment-friendly electricity. (AI generated illustration)
Author’s note: Everything you’ve read until now is technology news that you may already know, thanks to the internet. But there’s another story behind this article. Some time ago at Biggani.org, we interviewed Professor Jamal Uddin. As part of our interview project, we asked him to introduce us to a Bangladeshi scientist. He put us in touch with Dr. Shahiduzzaman Sohel. I reached out to Dr. Shahiduzzaman Sohel and collected three of his research papers. After reading those papers, I realized Dr. Sohel has been working tirelessly to solve this solar panel problem and has achieved some remarkable results. This article aims to present those findings in simple language for Bangla-speaking readers.
A New Hope: Perovskite
This is when a remarkable material was discovered—perovskite. This is a type of crystal that absorbs sunlight extremely well. Its chemistry is comparatively simple, and the most important thing is—it can be made into a thin film from liquid, meaning there’s no need for a large factory.
Perovskite solar cells are made from a combination of organic and inorganic materials—a blend known in scientific circles as the Hybrid Perovskite Solar Cell. Since the discovery of this technology, a new horizon has opened in solar energy research, as its efficiency has jumped from 3% to around 25% in a short time.
But here’s the catch: it’s not durable! While silicon solar cells can last up to 20 years, perovskite solar cells often degrade within weeks or months. The main reasons are air and moisture, temperature changes, and chemical breakdown due to light.
These materials are so sensitive that even a small amount of water vapor in the air can ruin their chemical structure. That’s why perovskite solar cells are made in extremely dry environments called “Glove Boxes”—airtight spaces where there’s no oxygen or humidity.
This makes it difficult to get this technology into the hands of ordinary people, as such complex setups are expensive to build.
There’s another issue: particle size and structure. The performance of perovskite solar cells greatly depends on their structure—how large the particles are, how evenly they are distributed, and whether there are gaps or holes between them. If the particles are small and uneven, it disrupts the flow of electricity; electrons get stuck, and the cell’s efficiency drops. On the other hand, if the particles are large and uniform, the cell can generate more electricity. Controlling the size and structure of these particles is difficult because perovskite is formed from a liquid solution that dries very quickly, causing crystals to form haphazardly.
Why Is It Important to Solve These Issues?
Scientists know that if these three major challenges—humidity, particle structure, and durability—can be solved, perovskite solar cells could revolutionize the world’s energy sector. Because it:
- Can be made with inexpensive materials,
- Is light and flexible,
- Can be used for everything from small mobile devices to large power stations,
- And can even be installed on fabric or plastic!
That’s why research on perovskite solar cells is being conducted in thousands of labs around the world today.
A New Direction for Scientists: Nanotechnology and Ionic Liquid
To solve these problems, scientists have turned to two innovative technologies—nanotechnology and ionic liquid.
Nanotechnology means controlling materials at the scale of extremely small particles called nanoparticles. The nanoscale refers to a scale so tiny that the size is less than one billionth of a meter (nanometer range).
At this scale, matter behaves differently than at normal sizes, resulting in new electrical, chemical, and mechanical properties. Light absorption, electron flow, even the color of materials changes. That’s why scientists are trying to control the structure of perovskite by breaking it down into tiny nanoparticles.
On the other hand, ionic liquid is a type of liquid that contains positive and negative ions but doesn’t easily dry or evaporate like regular liquids. Using this liquid to make perovskite allows the material to form slowly, resulting in well-shaped crystals. It creates a kind of “friendly environment” for perovskite so that it takes form properly and isn’t easily damaged by exposure to air.
Now let’s look at some of the instruments used in this field: AFM and Coating Technology. Scientists use some specialized tools in such precise research. For example:
AFM (Atomic Force Microscope): This is a special type of microscope that doesn’t use regular light or lenses, but instead uses a fine needle to touch every grain and bump on the surface of a material. This allows researchers to see how smooth a perovskite film is, where there may be defects, and the precise size of the particles.
Coating Technology: To make a perovskite film, the liquid is spread evenly using different techniques called “coating.” There are several types: Spin Coating—spinning the solution to spread it out; Bar Coating—spreading with a metal bar; Slip Coating—pouring the liquid on a slope and letting it flow down. This coating produces the thin layer of perovskite that later acts as the solar cell.
A Scientist’s Three-Stage Discovery
Science never takes leaps to make great discoveries. It advances step by step—what seems impossible today might be the beginning of a new path tomorrow. Here, a scientist and his research team have gradually solved the major problems of perovskite solar cells.
(1) Stage One: The Mystery of the Small Particle (2014 Research) The journey began with a simple yet profound question—does perovskite always have to exist as a single continuous sheet? Or, can it function if divided into tiny particles? This question led to research in 2015. The scientists broke down perovskite into nanoparticles—so small they’re invisible, even to standard microscopes. But there was a problem: perovskite dries very quickly, so the particles form into random shapes—sometimes elongated, sometimes ribbon-like, sometimes full of gaps. The researchers then used a special liquid—ionic liquid. This ionic liquid helped perovskite form slowly, resulting in small, almost spherical, and evenly distributed particles. In this study, the scientists showed for the first time that perovskite nanoparticles could be made using a simple spin coating technique. This was a fundamental breakthrough. The focus here wasn’t on electricity generation efficiency, but on answering the question: Can we control the structure of perovskite? The answer was “Yes.”
(2) Stage Two: The Battle to Survive in the Air (Research from 2021–2024) After the first stage was successful, an even bigger question remained—would this beautiful structure survive in the real world? Before, perovskite used to deteriorate rapidly in air, so it had to be made in closed rooms in the lab where air couldn’t get in. But in real life, solar cells have to endure open air. In this stage, the researchers again turned to ionic liquid, but in a new way. They found that ionic liquid not only helps make the particles but also forms an invisible shield over the perovskite. This shield prevents humidity and water from entering. In this research, bar coating and other new techniques were used—much like painting a wall with a metal rod to spread the liquid evenly. This allowed for the creation of larger perovskite films, crucial for manufacturing. The study showed that perovskite films made in regular air conditions lasted much longer and retained almost the same power output. This was a major success, as for the first time, scientists showed that it was possible to make perovskite outside of a glove box.
(3) Stage Three: Integrating It All (From Lab to Industry): 2025 The final study was the most mature step of this journey. Here, scientists combined the lessons of the previous two stages. This time, they didn’t just use perovskite nanoparticles as a test, but rather as “seeds” from which the whole perovskite film would gradually grow.

A consortium in Japan is currently testing the durability and performance of lightweight,
flexible perovskite solar modules at Osanbashi Pier, Yokohama.
This experimental system is installed in strong and salty wind conditions
to assess its effectiveness in harsh weather.
This outdoor trial is part of a larger three-year collaborative research project on perovskite solar technology.
Partner company Reiko has created these flexible perovskite solar modules,
which we are gradually moving towards commercial production.
This is part of an ongoing project by Japan’s Ministry of the Environment,
where mainly our perovskite seed technology has been used.
The entire project is being led by Professor Miyasaka of Toin University, Yokohama.
The Promise of the Future
These discoveries will help create solar cells in the future that can be placed on school roofs, village homes, even bags or clothing. They will be lightweight, affordable, and environmentally friendly. Above all, these studies deliver a message to Bangladeshi students—science isn’t just about memorizing books; science is about solving the world’s problems.
The Story of a Village Boy: Dr. Shahiduzzaman Sohel and the Journey of Scientific Dreams
The scientist behind all these endeavors is, in fact, from Bangladesh. Science doesn’t always start in big cities, modern labs, or with expensive equipment. Often, it begins in a small village home, where there’s sometimes no stable electricity, no internet, but an unstoppable curiosity and a hunger for knowledge. Dr. Shahiduzzaman Sohel’s story is just such a story. Growing up in a remote area of Bangladesh, Dr. Sohel’s childhood wasn’t easy. Surrounded by limitations—scarce resources, little access to education, almost no way to learn about modern science. And yet, there was a hidden power—the courage to ask questions. From an early age, he wanted to know, “Why does this happen?”, “How does it work?”, “What’s really inside?” That inquisitive mindset is what gradually leads a person down the path of becoming a scientist.

Dr. Shahiduzzaman Sohel on the left
and Prof Miyasaka on the right
(Pioneer of perovskite solar research), Osaka, Japan.
From Curiosity to Research—A Long Journey Science didn’t just appear in Dr. Shahiduzzaman Sohel’s life out of nowhere. It was the result of years of hard work, failures, and starting over. In his pursuit of new knowledge, he often faced research challenges where everyone around him said, “This is too difficult,” “Maybe this isn’t possible.” But a true scientist never runs from questions. Dr. Shahiduzzaman Sohel didn’t either. Perovskite solar cell, ionic liquid, nanoparticle—today these words sound simple, but at one point they were entirely new and uncertain research topics. Many of the answers were unknown even worldwide. He tried to understand—why does perovskite break down in air, how do you control the structure of the particles, and how can this technology be made practical for real life? This quest for understanding led him from one research project to another.

Dr. Shahiduzzaman Sohel and his colleagues
with Kanazawa University President Takashi Wada
(standing far left in the second row).
Three Research Projects, One Philosophy
The work of Dr. Shahiduzzaman Sohel and his team is not only a technological achievement but also reflects a philosophy: don’t fear problems—study and break them down patiently. In the first study, he learned how to control perovskite’s structure. In the second, he realized the challenge of durability in the real world. And in the third, he showed how to combine all this knowledge to craft powerful solutions for the future. This teaches us that major breakthroughs never happen in a day; they come from persistent thinking, learning from mistakes, and a never-give-up mindset.

Dr. Shahiduzzaman Sohel’s success is not just a personal achievement—it’s a matter of pride for Bangladesh. He has proven that you don’t have to be born in a developed country to become a world-class scientist; what you need is world-class thinking. Bangladeshi students can do world-class research if they learn to ask questions, patiently learn, and aren’t afraid of hard work.
The biggest lesson from Dr. Shahiduzzaman Sohel’s life is that science is not about exam scores or rote learning. Science is curiosity, observation, and the courage to exceed your limits. If you ask a question today, and fail ten times seeking the answer — you’re still on the scientist’s path. If you don’t understand today but try again tomorrow—you are still making progress. The greatest lesson from Dr. Sohel’s life is this: circumstances may limit you, but if your curiosity remains free, then your future will be free as well.
The research on perovskite solar cells being done today may one day solve even greater challenges—and maybe that scientist will come from a remote school in Bangladesh. So ask questions, be curious, don’t be afraid to make mistakes. Science begins the moment you say, “I want to understand this.” Dr. Shahiduzzaman Sohel’s story shows us that with hard work, curiosity, and a scientific mindset, world-class discoveries are possible from anywhere in the world.
References
1. Ionic liquid-assisted growth of methylammonium lead iodide spherical nanoparticles by a simple spin-coating method and photovoltaic properties of perovskite solar cells† Check for updates, M. Shahiduzzaman, Kohei Yamamoto, Yoshikazu Furumoto, Takayuki Kuwabara, Kohshin Takahashiab and Tetsuya Taima
2. ACS Applied Materials & Interfaces, Vol 13/Issue 18 “Ionic Liquid-Assisted MAPbI3 Nanoparticle-Seeded Growth for Efficient and Stable Perovskite Solar Cells” Md. Shahiduzzaman, LiangLe WangShoko, FukayaErsan, Y. MuslihAtsushi, KogoMasahiro Nakano, Makoto KarakawaKohshin, Takahashi, Koji Tomita, Jean-Michel Nunzi, Tsutomu Miyasaka, Tetsuya Taima
3. Solar Energy Materials and Solar Cells
4. Chemical Engineering Journal
affordablecarsales.co.nz












Leave a comment